Can Artificial Intelligence Predict Breast Cancer?
Research studies have shown that leveraging artificial intelligence algorithms to analyze mammogram images can help determine a person’s risk for breast cancer.
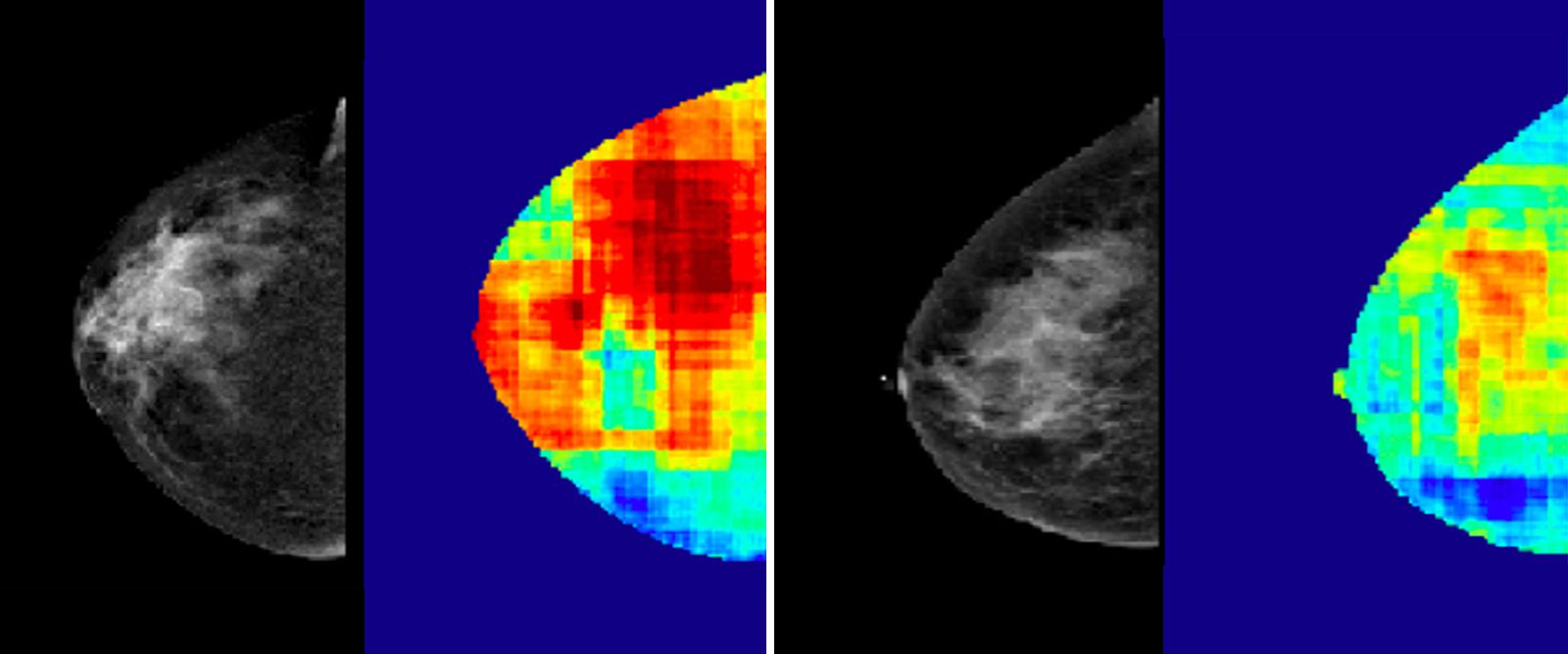
Using mammogram images of two patients who have similar breast densities, an artificial intelligence algorithm detected regions that represent high cancer risk (in red).
Breast cancer is the most common form of cancer in the world. In the United States, women have a one in eight chance of being diagnosed with breast cancer in their lifetime, according to the American Cancer Society (ACS). It is also the second-leading cause of cancer death in women.
Over the years, experts have developed various tools and technology that support screening, detection, and treatments to improve outcomes for this deadly and common disease. But what if doctors had a way to accurately predict the likelihood of someone developing breast cancer? Could prediction change the landscape for breast cancer care and survival rates?
Experts believe it can, and the development of risk prediction tools is underway thanks to artificial intelligence (AI). Through analyzing large amounts of data in mammogram images, deep learning — a form of AI — has been found to more accurately predict the risk of breast cancer occurrence, says Dr. Katia Dodelzon, associate attending radiologist at NewYork-Presbyterian/Weill Cornell Medical Center.
Understanding AI
According to the National Cancer Institute, implementing AI in cancer care could improve the accuracy and speed of diagnosis, aid in clinical decision-making, and lead to better health outcomes. The institute defines AI, its types, and subtypes, including deep learning, as:
- AI: “a computer performing tasks commonly associated with human intelligence. Humans code or program a computer to act, reason, and learn. An algorithm or model is the code that tells the computer how to act, reason, and learn.”
- Machine learning: “a type of AI that can learn to make predictions or decisions. The more data a machine learning model is exposed to, the better it performs over time.”
- Deep learning: “a subset of machine learning that uses artificial neural networks. A well-designed and well-trained deep learning model can perform classification tasks and make predictions with high accuracy.”
Health Matters spoke with Dr. Dodelzon to understand how these AI models work, the difference they can make in breast cancer prevention and screening, and the future of utilizing AI in breast cancer care.

Dr. Katia Dodelzon
How do mammogram images help AI models predict breast cancer?
Dr. Dodelzon: Mammogram images are widely accessible and are immense reservoirs of data. The images contain a wealth of information, such as pixels that can signify patterns, and allow breast radiologists to detect breast cancer and assess breast density. Other than this, there is much more information in mammograms that while not perceptible to the human eye, it is to AI including deep-learning models. These algorithms can quickly detect and analyze their patterns for potential breast cancer risk.
Why are risk-prediction tools powered by AI important when it comes to a disease like breast cancer?
Most breast cancers are seen in women without easily identifiable predisposing risk factors like genetic mutations, such as BRCA 1 and BRCA 2, or who have a personal or significant family history.
To tackle the growing breast cancer burden, early detection with screening is vital. We know that diagnosing breast cancer at a smaller size in an early stage, as is accomplished by screening, leads to significant improvement, not just in mortality (so more lives are saved) but also in decreasing morbidity from treatment. A smaller cancer requires a smaller or less invasive surgery, for example. So, the key is risk-level appropriate screening.
There are multiple tests, such as the Tyrer-Cuzick, Gail, and Breast Cancer Surveillance Consortium models, that are currently available, clinically, to calculate a woman’s risk. They are questionnaires that calculate a woman’s risk score based on answers such as their family history of the disease and breast density. The problem with all these calculators is that they can underestimate risk in some women. It is important to have accurate risk-prediction tools because it can help guide screening and prevention strategies that can ultimately improve the outcome of patients.
How do deep learning models utilize mammogram images to make risk predictions?
Once we input mammograms into the model, it analyzes the data — the variations in pixels — and provides a score for future risk of breast cancer, based on the data and textural features that it sees in the images. We then provide the model feedback, such as ‘You were correct. This woman did develop breast cancer,’ thus further validating the risk score that it generated. We train the algorithm with mammograms of women whose outcomes we know (whether they ended up developing breast cancer or not). This allows us to continually strengthen and improve the accuracy of the algorithm.
What is the difference between utilizing AI for breast cancer risk prediction and detection?
The power of AI is largely in “seeing” things that we humans cannot, such as its ability to extract and analyze additional image-based information to aid in the prognosis and prediction of breast cancer. It takes breast imaging a step further to inform personalized screening and treatment approaches more precisely. It is a tool that arms us with the ability to do more for our patients.
AI is being studied for actual breast cancer diagnosis, but these studies are limited and the outcomes are variable, as there are limitations to the inherent image quality and technique, and cancer detection is a complex task. Overall, the best performances are seen when combining interpretations from breast imaging radiologists and physicians with those of artificial intelligence. With further studies and improvements, there is potential of this is in places where there are fewer expert breast imagers. It can help augment interpretative skills to a larger patient population and decrease disparities in access.
What impact can the deep learning risk-prediction model make?
Ultimately, deep learning risk prediction tools can help people who are disproportionately impacted by breast cancer. Incidence and death rates differ substantially by race and ethnicity: breast cancer occurs more often among white women, but more Black women die from the disease — surpassing deaths from breast cancer in white women by 40%.
When the model is further validated, this risk assessment tool has the potential to be implemented in clinical settings and applied individually to each patient’s mammogram. This can help provide individual breast cancer risk prediction scores, which can inform and tailor individual screening strategies and more accurately identify high-risk women who may benefit from earlier and more frequent screening.
What are the next steps?
At NewYork-Presbyterian/Columbia University Irving Medical Center and NewYork-Presbyterian/Weill Cornell Medical Center, we are in the process of conducting more research and exposing the deep learning model to a greater diversity of patient populations. We are working on expanding this study to continue to validate, build, and perfect the model. Because it is a deep learning algorithm, it is always learning and can continually improve. The more feedback we give it, the better it is going to perform.
Katia Dodelzon, M.D., is a board-certified radiologist specializing in breast imaging and an assistant attending radiologist at NewYork-Presbyterian/Weill Cornell Medical Center. She is vice chair of Clinical Operations for the Department of Radiology, an associate professor of clinical radiology, and associate program director of the Breast Imaging Fellowship Training Program at Weill Cornell Medicine.

