Breast Density Information to Become a Regular Part of Mammogram Results, the FDA Announces
A radiologist at NewYork-Presbyterian Westchester shares what to know about the new requirement to inform patients if they have dense breast tissue, a risk factor for breast cancer.
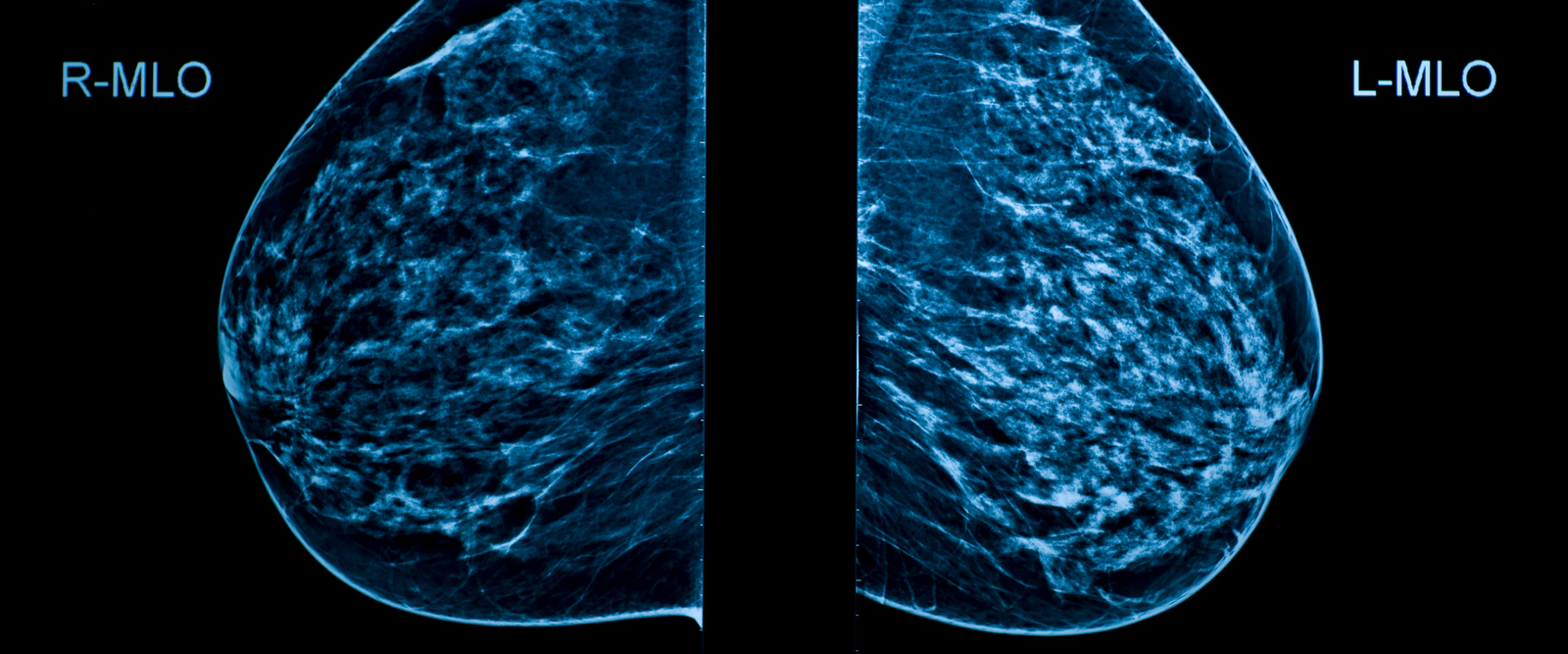
People receiving their mammograms will now be notified of their breast density, a risk factor for breast cancer, according to new requirements from the U.S. Food and Drug Administration (FDA). “In addition to receiving a notification that their results are benign, or negative, patients will receive notification that they have dense breast tissue or that they do not have dense breast tissue,” says Dr. Lauren Friedlander, a radiologist at NewYork-Presbyterian Westchester, who shared what to know about the updates and why they are important.
In March 2023 the FDA had first announced the requirements. At the time, 38 states had already passed laws requiring mammogram centers to tell women their breast density status. States that did not have these requirements in place had 18 months to implement them.

Dr. Lauren Friedlander
Because dense breast tissue can make it difficult to detect breast cancer on mammograms, the FDA is advising people to speak with their medical provider once they are informed of their status. Around half of women 40 years or older have dense breast tissue, according to the Centers for Disease Control and Prevention.
In the new updates from the FDA, “there’s language that lets patients know that women who do have dense breast tissue may benefit from additional screening exams [such as ultrasounds or MRIs],” says Dr. Friedlander. “That’s important because it really promotes a conversation between a patient and her physician about how best to optimize early cancer detection.”
Breast cancer is the most common cancer in women in the U.S., and it is about 30% of all new female cancers each year, according to the American Cancer Society. Overall, there is a one in eight chance that a woman will develop breast cancer in her lifetime.
“Mammograms continue to be the best tool for breast cancer screening and detection,” adds Dr. Friedlander. “It is the mainstay of breast cancer screening, the only test that we have that has a proven mortality benefit that’s been studied for many, many years.”
Additional Resources
Find out more about breast cancer screenings.
Learn about breast cancer care at NewYork-Presbyterian.

