Are Absorbable Stents the Future of Cardiac Care?
A new angioplasty technology undergoing testing may lead to fewer repeat procedures to keep arteries clear.
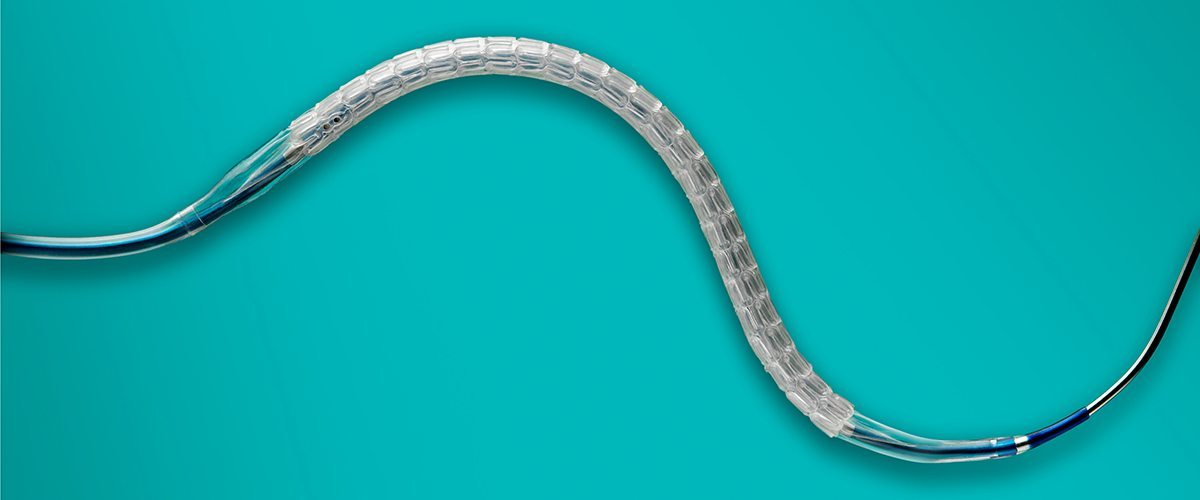

Some 735,000 Americans suffer a heart attack each year, according to the Centers for Disease Control and Prevention. For those who survive, the traditional form of treatment is angioplasty, in which a small metal mesh or scaffold is inserted into the blocked artery to widen the vessel and restore blood flow. It is also commonly used to treat coronary artery disease and angina.
Though these devices, called stents, do their job within six to nine months, they remain with the patient for the rest of his or her life. Sometimes, scar tissue forms within the stent, causing the artery to narrow again and necessitating a repeat procedure.
“There’s always been an interest in trying to develop technology that serves the patient during the time that you need it to, but then goes away when you don’t need it to be there any further,” says Dr. Ajay Kirtane, interventional cardiologist and director of cardiac catheterization laboratories at NewYork-Presbyterian/Columbia University Medical Center. “Unfortunately, the technology hasn’t been there.”
But this may be about to change. A new device called Absorb stent from Abbott Vascular was approved in 2016 by the U.S. Food and Drug Administration after a yearlong study of 2,008 patients found that it was comparably safe to traditional metal stents. The device releases a drug called everolimus that limits the growth of scar tissue; the underlying scaffold is designed to fully dissolve into the body after about three years. Four small metal markers, visible by X-ray, are left behind to show where the scaffold was. Absorb’s approval has some doctors cautiously optimistic about its use in patients.
“Those are some of the physical differences; otherwise, it’s got the same drug [everolimus] on it, which releases at the same rate, and it seems to be just as effective,” says Dr. Gregg W. Stone, global chairman of the study and director of cardiovascular research and education at the Center for Interventional Vascular Therapy at NewYork-Presbyterian/Columbia University Medical Center. “The big difference then, is that after several years, it completely disappears, whereas metal stents stay there forever.”
Absorb is made of a plastic-like polymer and works much like surgical sutures that dissolve. The possible advantages, according to Dr. Stone, who led the national, multi-center study, are a reduced need for repeat procedures after the stent has dissolved and fewer risks that can accompany traditional metal stents.
Health Matters spoke with Dr. Stone about the risks and benefits of Absorb, why more testing is needed and the factors every patient should consider.
Though the FDA approved Absorb, more testing is being done. Why?
Dr. Stone: The hope is that it reduces the need for repeat procedures, but we don’t know that yet. What we’ve learned is that regular metal stents can fail at a rate of about 2 percent per year after the first year — that’s about one in 50 patients every year. The arteries could re-narrow or blood clots could form in them. A lot of patients will have this device for 10 or 20 years, and when you add up all those years, it can be a rather high failure rate. We think that is caused by the stent being a permanent fixture in the body. We performed the largest study of the device, which led to its approval in the United States. The next study, which is even larger, is designed to determine whether this device is superior to standard permanent metal drug-eluting stents.
How is having the Absorb stent implanted different from a metal stent?
The procedure may be 10 to 15 minutes longer. Because the polymer [Absorb] stent is a little thicker, doctors have to pay more attention to how it’s implanted. The metallic stents are a little bit more forgiving. Recovery is the same.
Who is the ideal candidate for Absorb?
Right now, these devices are meant for patients who are relatively low risk, and with relatively noncomplex blockages. We are looking for patients with lesions that are somewhat soft, and with mostly large vessels. For a younger patient, typically age 65 or younger, with big arteries, and a lot of heart muscle, these devices can be really optimal.
Who is the ideal candidate to implant the device? Not every doctor has done this yet.
It’s not that different than a regular stent, but if a doctor says they’ve implanted 25 or more, I would say that’s very good experience. Then you might ask, “I heard this takes a little more time to do a really good job implanting it. Do you agree with that?” If they agree, that would make me feel good. If they say, “Eh, there’s nothing to it,” then I wouldn’t be quite as comfortable. Your doctor needs to be willing to take the extra 10–15 minutes to do a really good job. That doesn’t sound like much. However, it’s a crucial step in ensuring the implant is successful.

