What Is Precision Medicine?
Dr. Gary Schwartz explains this major innovation in how doctors diagnose and treat cancer patients.
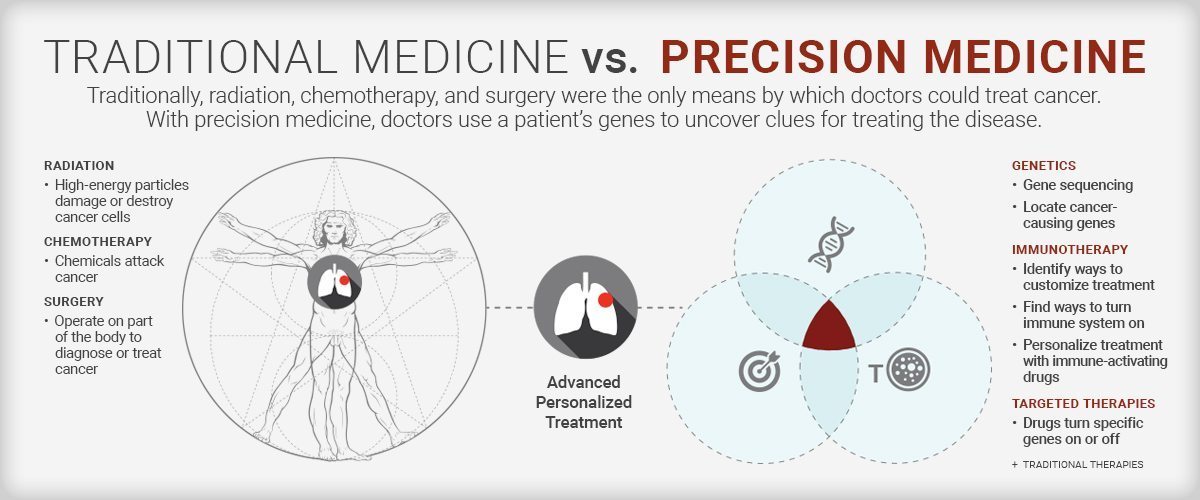

Today’s oncologists are no longer treating cancer with a one-size-fits-all approach. They’re using precision medicine: a strategy in which the patients’ genes provide the clues to potentially defeating their cancer. At NewYork-Presbyterian and its academic partners, Columbia University Irving Medical Center and Weill Cornell Medicine, clinicians and scientists are using next-generation sequencing tools to gain insight into each patient’s disease. We spoke with Dr. Gary Schwartz, chief of the hematology and oncology division at NewYork-Presbyterian/Columbia University Irving Medical Center to understand how treating cancer at the genomic level is having a profound impact on cancer care.
What is the precision medicine approach to treating cancer?
With precision medicine, we are now in the position to make treatment decisions not simply based on the specific cancer but on the genes that make up that cancer. By examining the DNA of a patient’s tumor, we can identify the cancer-causing genes that make cancers grow. With this information we have the ability to find a drug that targets that gene, so we can turn the gene off and attack the cancer cell.
Can you explain how precision medicine leads us to new therapies for patients?
A good example of the progress made in outsmarting cancer with precision medicine can be seen in melanoma. We know now there is one specific gene that is turned on by the sun and sunburn. It’s called B-RAF. This gene is mutated in over 60 percent of patients with melanoma. Where we didn’t have treatment options before, we now we have at least two drugs — vemurafenib and dabrafenib — [that] block B-RAF and stop the cancer from growing and spreading.
How has DNA sequencing allowed doctors to provide personalized cancer care? And how has it evolved over the years?
With the capability to now sequence DNA, we can tell the patient which genes turn cancer on and which genes turn cancer off. We can do this for every person’s cancer. This allows us to understand at a genomic level what is fueling a patient’s cancer to grow. Twenty years ago, gene sequencing cost millions and took months to receive results. With current technology at our cancer centers, we can do the sequencing and the interpretation of the sequencing at a reasonable cost and get this information back to the patient and the patient’s doctor within a few weeks.
How long does the process take for a patient and what does it entail?
We have a group of specialists who examine [a patient’s individual] gene patterns and then make treatment recommendations based on what is known about the abnormal genes and whether drugs exist to block them. We also can make recommendations for clinical trials based on this information, many of which are being conducted at NewYork-Presbyterian. The actual sequencing of the DNA now takes only seconds. The complexity comes from the “reading” of the DNA and identifying the abnormal genes. This is what still takes time and can take up to four weeks to analyze. But with more sophisticated computers even this timeframe is shortening. I anticipate that as the technology improves we should also have this down to several days.
“With the capability to now sequence DNA, we can tell the patient which genes turn cancer on and which genes turn cancer off.”— Dr. Gary Schwartz
What role does precision medicine play in treating cancer with immunotherapies?
In immunotherapy we find ways to turn on the patient’s immune system to fight their cancer. With this approach we are seeing cures in certain cancers (like melanoma) where the use of the word “cure” was not even possible five to 10 years ago. We are now working to understand which patients most benefit from immune-activating drugs, then our goal is to “personalize” a treatment for them. Though this is being used to some degree in current clinical practice, our investigators are fast at work trying to define new biomarkers to predict response in both the patient’s cancer and even in the immune cells that circulate in the patient’s blood.

Dr. Gary Schwartz
How does precision medicine help identify who could benefit from immunotherapy treatment?
Precision medicine allows us to better identify the treatment from which the patient will receive the most benefit. The idea is to essentially take the “guesswork” out of drug selection. With precision medicine, we can now begin to understand who will or who will not respond to immunotherapy. It allows us to identify the best drug treatments early on and puts the patient more quickly on the path to potential cancer cure while minimizing side effects.
What are some of the national initiatives using precision oncology?
One study advancing precision medicine is the NCI-MATCH trial led by the National Cancer Institute. This clinical trial analyzes a patient’s tumors to determine if they contain gene abnormalities for which a targeted drug exists and assign treatment based on the abnormality. The trial, which is open to patients at both our cancer centers, is still enrolling patients and has been expanded to include 7,000 cancer patients in the United States.
There are other national studies open at cancer centers across the country for precision medicine and we encourage participation in clinical trials. This includes the American Society of Clinical Oncology (ASCO) program called Targeted Agent and Profiling Utilization Registry (TAPUR).
The national cooperative groups for various cancers are also involved in this process. Gene sequencing is particularly important in rare cancers where treatment options are limited. For those patients gene sequencing can provide unique insights for cancer therapy.
Watch a quick video on how precision medicine is cracking the code on cancer care.

